FDA Authorizes Moderna and Pfizer-BioNTech COVID-19 Vaccines for Children Down to 6 Months of Age
Today, the U.S. Food and Drug Administration authorized emergency use of the Moderna COVID-19 Vaccine and the Pfizer-BioNTech COVID-19 Vaccine for the prevention of COVID-19 to include use in children down to 6 months of age.
- For the Moderna COVID-19 Vaccine, the FDA amended the emergency use authorization (EUA) to include use of the vaccine in individuals 6 months through 17 years of age. The vaccine had been authorized for use in adults 18 years of age and older.
- For the Pfizer-BioNTech COVID-19 Vaccine, the FDA amended the EUA to include use of the vaccine in individuals 6 months through 4 years of age. The vaccine had been authorized for use in individuals 5 years of age and older.
>NYC Activists plead with parents not to get their kids vaccinated<
Related – Pfizer Seeks Approval to Inject Infants with Deadly Gene Therapy Shots
CDC With a smile encouraging parents to sentence their children to ill-health and potential death
Ryan Cloe, MD – Crazy and criminal to give the Covid vaccine to babies
Pfizer’s Claim That the Jab Was Safe for Pregnant Women Was Based On a Biased French Rat Study
A Shot in the Dark Documentary (Before COVID, Bad Baby Vaccines) That we were all given….
Massive Bio-Structures Found in Bodies of the Vaccinated
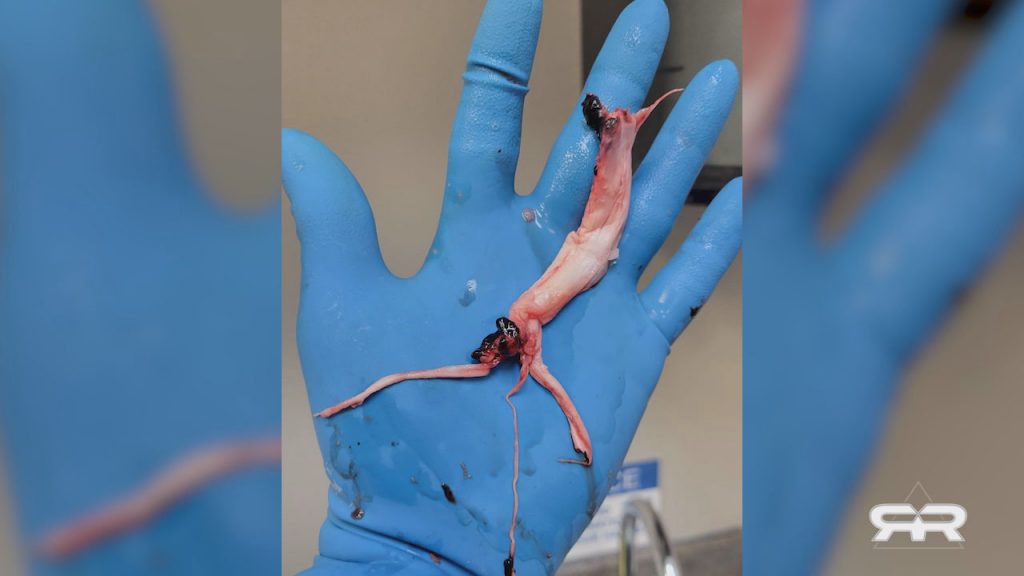
>NYC Activists plead with parents not to get their kids vaccinated<
UNITED STATES UNDER >MILITARY CONTROL<
>>Restored Republic<<
Active Executive Orders and [National Emergencies] listed
WHAT ARE WE FIGHTING WORLDWIDE?
THE FALL OF THE CABAL
>>The Birth of the Cabal<<

Ascension All Connected and Also Important

Ascension All Connected and Also Important